Generating Insights into Care Gaps and Fracture Risk in Patients with Osteopenia and Osteoporosis

Electronic Health Records, Real-World Data, and Natural Language Processing
Osteoporosis has long been recognized as a significant public health concern, yet this disorder of bone remodeling and heightened fracture susceptibility remains underdiagnosed and undertreated. Additionally, the incidence of osteoporosis and related fractures has been increasing alongside a burgeoning elderly population.1 These observations have warranted urgent action on the part of providers and health systems, with healthcare stakeholders closely evaluating osteoporosis and its consequences, its societal costs, and the measures needed to close ongoing gaps in care.
Osteoporosis and Fracture Risk
Fully half of Americans aged 50 years or older are estimated to have osteopenia (low bone mineral density) or osteoporosis (more severe bone loss). Each of these individuals has an elevated risk of bone fracture.
- Osteoporosis is associated with 2 million fractures annually in the United States. Such fractures cost the U.S. economy an estimated $19 billion in direct spending. By 2025, absent improvements in patient care, 3 million fractures attributable to osteoporosis are anticipated to cost over $25 billion.
- Of those who suffer fractures, most are postmenopausal women. Fracture incidence for women exceeds that of breast cancer, heart attack, and stroke combined. Fractures generally occur later in life for men than for women, with men more likely to die within a year after breaking a hip.
- Osteoporosis is a major risk factor for fragility fractures—fractures caused by low-trauma impact, such as a fall from standing height.
- Individuals who experience bone fractures have an increased risk for subsequent fractures.
- Nearly 80% of individuals who suffer fractures are not being tested or treated for bone loss.2,3
Although fractures occur more frequently beyond the seventh decade of living, advanced age is associated with lower use of fracture-preventive therapies.4,5 Fractures in the elderly increase the risk of morbidity, functional decline, disability, residential institutionalization, and mortality.4,6
Bone Mineral Density Testing and Diagnosis
As a measure of fragility fracture risk, bone mineral density (BMD) testing is the single diagnostic procedure capable of detecting osteoporosis in advance of a fracture.7 BMD testing may be used to reveal the presence of osteoporosis before or after a fracture; predict the likelihood of future fracture; indicate whether bone density is improving, worsening, or similar to that shown in the most recent prior test; and demonstrate the effectiveness of osteoporosis medications.8
Typically measured using dual energy x-ray absorptiometry (DXA), a patient’s bone density is usually compared with that of a 30-year-old healthy adult. The result is reported as a T- score—the standard deviation (SD) between the patient’s bone density and that of the healthy control—which may be used to predict the likelihood of a future fracture:
- A T-score of -2.5 or less indicates osteoporosis
- A T-score between -1 and -2.5 indicates osteopenia4,8
Fracture risk may also be evaluated with the Fracture Risk Assessment Tool (FRAX Tool). FRAX estimates an individual’s 10-year risk of a fragility fracture based on a small set of demographic, family history, and clinical characteristics. In addition, FRAX scores can help identify individuals who might benefit from taking preventive action or an osteoporosis medication.6
Fracture-Preventive Treatment
Delaying and preventing first or subsequent fractures is the primary goal of osteoporosis treatment. As a chronic condition, osteoporosis requires long-term, multi-modal management.
Treatments approved by the Food and Drug Administration (FDA) that prevent bone density loss include agents that destroy or reduce activity of bone degrading cells. These include the bisphosphonates, selective estrogen receptor modulators (SERMS), and a biologic (monoclonal antibody). Other agents increase activity of bone forming cells (i.e., parathyroid hormone [PTH] and PTH analogs and a biologic [monoclonal antibody]).9,10
Real-World Studies in Osteoporosis
Real-world data (RWD) include the comprehensive, health-related patient information collected from diverse populations during the routine delivery of medical care. Widely captured within electronic health records (EHR), medical claims and billing records, and other sources, RWD may be used to generate real-world evidence (RWE), defined as the clinical evidence regarding the use and potential benefits or risks of medical products.11 Both RWD and RWE have the potential to support clinical and regulatory decision-making and to inform drug and biologic development. For patients with osteoporosis, RWD and RWE may offer insight into the challenges and opportunities associated with its diagnosis, treatment, and long-term management.
Real-world studies in osteoporosis have evaluated, among other topics, therapeutic effectiveness of FDA-approved medications, fragility fracture risk and treatment gaps, and treatment adherence and/or persistence:
- A large, population-based study reported a decrease in the incidence of clinical fractures following initiation of osteoporosis therapies from several drug classes, in alignment with results from randomized clinical trials.12
- Four observational studies have demonstrated clinical effectiveness of a PTH agonist in mostly female patients with osteoporosis. Rates of fracture were shown to decrease by as much as 62%.13
- In a study of osteoporosis management in primary care, over three-quarters of female patients aged 70 years and older were not being treated for osteoporosis, despite having elevated fragility fracture risk.14
- Low or moderate adherence to osteoporosis medications was associated with a greater risk of experiencing a secondary fracture than high adherence to medications in women enrolled in Medicare with a previous fragility fracture.15
- A systematic review of real-world studies concluded that adherence to oral bisphosphonates was suboptimal overall.16
Recently, a real-world study commissioned by the National Osteoporosis Foundation (NOF) examined the clinical and economic impact of new osteoporotic fractures suffered by Americans who were enrolled in the Medicare fee-for-service program during calendar year 2015.17 The study, which leveraged de-identified medical claims data, reported several key findings:
- Two million Medicare beneficiaries suffered 2.3 million fractures.
- A total of 307,000 beneficiaries suffered one or more additional fractures at a cost of over $6.3 billion.
- One-half of secondary fractures might be avoided with appropriate treatment.
- Preventing repeat fractures by 20% could reduce costs billed to Medicare by $1.2 billion.
- Only 9% of patients underwent BMD testing within six months of a new fracture.
Retrospective Cohort Analysis
We conducted a retrospective cohort analysis to explore BMD assessment in adult ambulatory patients with or without a diagnosis of osteopenia or osteoporosis based on ICD-9-CM and ICD-10-CM codes. The analysis used RWD sourced from an EHR platform offered by Veradigm®.
RWD were derived from de identified patient records captured during an eight-year, three-month period (March 17th, 2009 to June 18th, 2017). RWD were used to generate RWE for insights into the patient cohorts and potential care gaps.
Preliminary analyses were first conducted using Evalytica, a Veradigm offering.
- Evalytica is a software-as-a-service (SaaS) application that enables transparent and efficient analysis of EHR, registry, and administration claims data that are available from diverse sources.18
The Evalytica analyses identified over 200,000 individuals with a diagnosis of osteopenia, osteoporosis, or other disorders of bone density and structure, with at least one prescription for or documented use of osteoporosis medications. Most diagnosed patients who were on medication were taking bisphosphonates (81.9%), followed by a biologic (12.4%), SERMS (9.8%), and PTH or PTH analogs (2.9%).
Additional analytics revealed 10,064 patients with (n=8,789) or without (n=1,275) a diagnosis of osteopenia or osteoporosis had completed DXA scans, with at least one T-score recorded. Because DXA results were mainly entered by physicians into Subjective, Objective, Assessment, and Plan (SOAP) notes, natural language processing (NLP) techniques were used to identify, extract, and present the data in a structured format. DXA imaging results recorded by physicians consisted of the anatomy scanned (i.e., spine, forearms, or bones in the hip), the standardized score, and the score type.19
- In the present analysis, 81% of T-scores were extracted from semi-structured and unstructured laboratory result descriptions using a machine learning-based NLP algorithm.
Most patients (92.3%) with T-scores were 65 years of age or older. Nearly one-quarter (24.5%) of patients with T scores were aged 80 years or older. Most patients (88.8%) with T-scores were female.
The percentages of patients with T-scores indicative of normal, osteopenic, or osteoporotic bone density are shown in Figure 1. For patients with a diagnosis, 39.4% had T scores indicative of osteopenia and 44.7% had T-scores corresponding to osteoporosis. For patients without a diagnosis, 44.2% and 13.7% had T-scores indicative of osteopenia and osteoporosis, respectively.
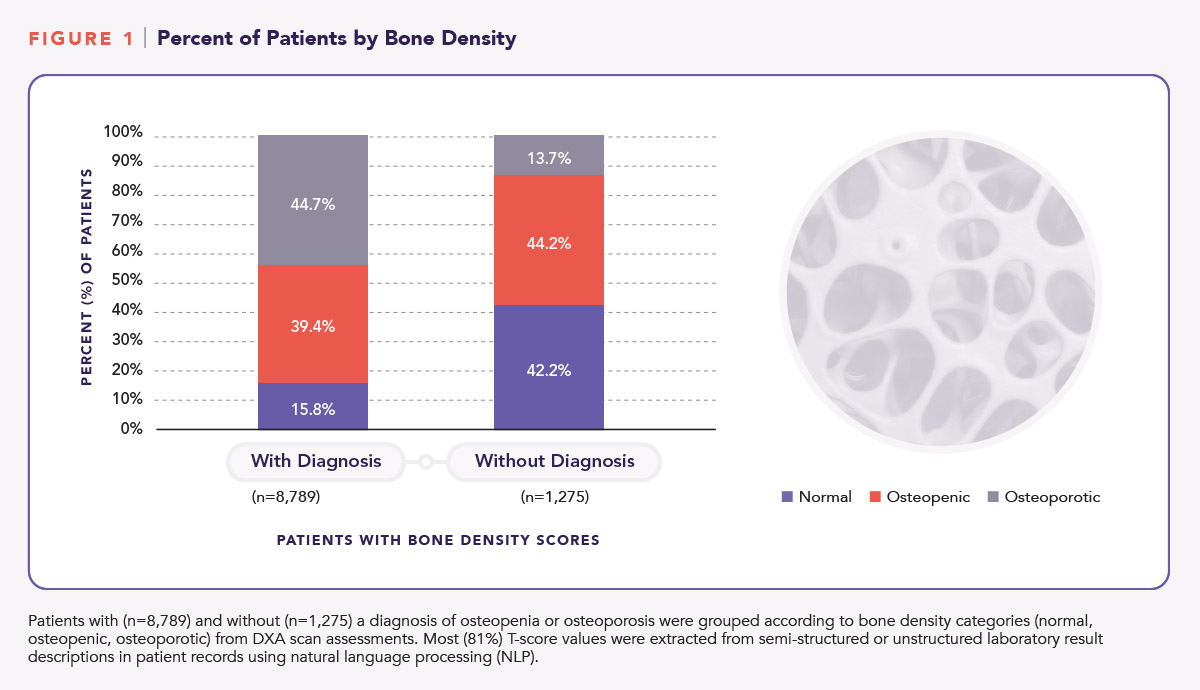
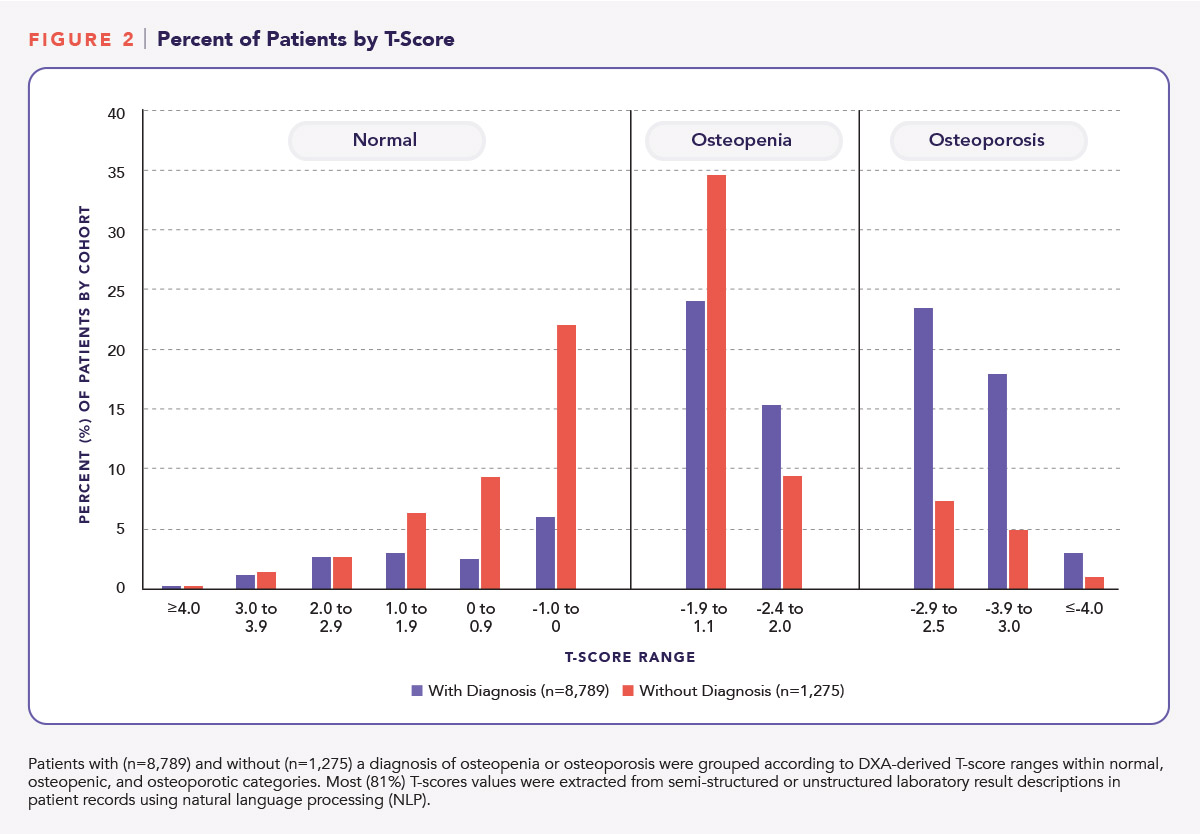
Figure 2 shows the percentage of patients with or without a diagnosis of osteopenia or osteoporosis according to T-score ranges. Twenty-one percent (21.1%) of patients with a diagnosis had recorded T scores of -3.0 or less; 6.1% of patients without a diagnosis had T-scores of -3.0 or less.
What clinical insights may be derived from the retrospective cohort analysis? Even as HCPs work toward meeting therapeutic needs and improving outcomes, care gaps remain for a significant number of patients with osteopenia and osteoporosis. Our analysis of de-identified patient data sourced from a Veradigm EHR revealed potential gaps in care for patients at risk of bone fracture.
- More than one half of patients without a diagnosis for osteopenia or osteoporosis had recorded DXA scores suggestive of osteopenia or osteoporosis.
- Fully one-fifth of patients with a diagnosis and 6% of patients without a diagnosis had recorded T-scores of -3.0 or less.
It should be noted that for some patients in this example analysis, diagnoses may have been recorded in a narrative format in unstructured data fields rather than as diagnostic codes captured in structured fields. NLP could be applied to extract narrative evidence for diagnoses or lack thereof. It is also possible that some diagnoses were not carried over from a previous or specialty care practice.
Patients with very low DXA scores (i.e., < -3.0) have an “especially high risk” of future bone fractures and are candidates for combination or sequential anti-fracture therapy.20 Fracture incidence has been shown to decline when approved pharmacotherapies are prescribed, irrespective of whether the patient has a T score in the osteopenic or osteoporotic range.20
The analysis also showed over 90% of patients with T-scores were 65 years of age or older, with nearly one-quarter of patients with T-scores aged 80 years or older. A cross-sectional study conducted in eight countries reported a high percentage (>75%) of female patients aged 70 years and older were not being treated for osteoporosis in the primary care setting, despite having elevated fragility fracture risk.14
The NOF recommends a comprehensive approach to diagnosis and management of fracture risk that leverages patient histories, physical examination findings, imaging to diagnose vertebral and other types of fractures, 10 year estimated fracture probabilities when available, and BMD/DXA assessments. The NOF considers BMD/DXA testing to be a reliable predictor of future fracture risk, with risk of fracture increasing exponentially as bone density decreases.20
Future Directions
How can information technology tools contribute to understanding and addressing care gaps and fracture risk in osteoporosis? As overlapping subfields of artificial intelligence, natural language processing, machine learning, and deep learning are digital capabilities with the potential for supporting healthcare delivery and for providing timely clinical insights into high-risk populations by curating, qualifying, and exploring the full value of mass medical data. As demonstrated in the current analysis, when DXA scan results are recorded in narrative formats within unstructured fields in EHRs, natural language processing may enable consistent and efficient background T-score identification, extraction, and presentation. Veradigm is working towards adoption of artificial intelligence and related technologies for managing RWD available through its EHR platforms and registries.
NLP also has the potential to translate difficult-to-understand medical records and other informational content for availability on patient-facing portals, in support of healthcare literacy, shared decision-making, and patient self-care.21 Among interoperative platforms, information technology tools may be used to provide insight into closing care gaps and to inform quality improvement and value-based care.22 Life sciences stakeholders are currently leveraging real-world retrospective data and artificial intelligence tools in an effort to accurately predict osteoporotic fracture onset.23,24
How might EHRs be used to enable or inform diagnosis and management of fracture risk in osteoporosis? EHRs are positioned to support HCPs in diagnosing and managing osteopenia and osteoporosis, as these platforms securely capture and document comprehensive and relevant, point-of-care patient data. Within the clinical workflow, cloud-based interactive EHRs may offer evidence-based tools that enable identification of patients at high risk of bone fracture. For example, EHR tools may support HCPs in scheduling DXA scans for diagnostic purposes, for monitoring bone loss longitudinally once a diagnosis is made and treatment has begun, and for assessing risk of a repeat fracture when a primary fracture has occurred. Together with clinical variables that are critical to an understanding of fracture risk—e.g., age, gender, comorbidities, fall history, and polypharmacy—DXA scan findings may be leveraged to inform guideline-based, individualized treatment plans that facilitate provider-patient discussions and align with each patient’s ability to make informed choices regarding his or her care.
Suboptimal medication adherence and persistence are common among patients with osteoporosis. EHR support tools may afford access to educational content that elevates health literacy, so patients are better informed regarding bone health, modifiable risk factors, and the value of osteoporosis medications. Educational materials may be shared by HCPs at the point of care or through linked patient–provider portals, both mobile- and web-based. By providing access to informational material, EHRs may support HCPs in enabling patients to fully engage as partners in their own health and well being. To the extent that analysis of RWE provides insights into clinical inertia or patient reluctance to manage fracture risk, life sciences stakeholders may consider development solutions, product life-cycle innovations, or commercial strategies to improve bone care for aging Americans.
For more information on datasets and analytic services offered by Veradigm®, contact us.
References:
1 Tarantino U, Cannata G, Celi M, et al. Incidence of fragility fractures. Aging Clin Exp Res 2007;19(4 Suppl): 7-11.
2 NOF 2019. National Osteoporosis Foundation. Osteoporosis Fast Facts. https://cdn.nof.org/wp-content/uploads/2015/12/Osteoporosis-Fast-Facts.pdf Accessed 18 November 2019.
3 NOF 2019. National Osteoporosis Foundation. Just for men. https://www.nof.org/preventing-fractures/general-facts/just-for-men/
Accessed 15 December 2019
4 Berry SD, Kiel DP, Colón-Emeric C. Considering the risks and benefits of osteoporosis treatment in older adults. JAMA 2019;321(22):2231-2233.
5 Ensrud KE, Kats AM, Boyd CM, et al. Association of disease definition, comorbidity burden, and prognosis with hips fracture probability among late-life women. JAMA Intern Med 2019:179(8):1095-1103. doi:10.1001/jamainternmed.2019.0682.
6 Friedman SM, Mendelson DA. Epidemiology of fragility fractures. doi: 10.1016/jcger.2014.01.001.
7 Compston JE, McClung MR, Leslie WD. Osteoporosis. Lancet 2019;393:364-376.
8 NOF 2019. National Osteoporosis Foundation. Bone density exam/testing. https://www.nof.org/patients/diagnosis-information/bone-density-examtesting/ Accessed 18 November 2019.
9 Eastell R, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2019;104:1595-1622.
10 Langdahl BL, Andersen JD. Treatment of osteoporosis: unmet needs and emerging solutions. J Bone Metab 2018a;25(3):133-140.
11 FDA.gov 2019. https://www.fda.gov/science-research/science-and-research-special-topics/real-world-evidence
Accessed 24 November 2019.
12 Yusuf AA, Cummings SR, Watts NB, et al. Real-world effectiveness of osteoporosis therapies for fracture reduction in post-menopausal women. Archives of Osteoporosis 2018;13:33. doi.org/10.1007/1657-018-0439-3.
13 Langdahl BL, Silverman S. Fujiwara S, et al. Real-world effectiveness of teriparatide on fracture reduction in patients with osteoporosis and comorbidities or risk factors for fractures: Integrated analysis of 4 prospective observational studies. Bone 2018b;116:58-66.
14 Amgen 2019. Amgen Presents New Data At WCO-IOF-ESCEO 2019 Revealing Osteoporosis Treatment Gap In Europe.
https://www.amgen.com/media/news-releases/2019/04/amgen-presents-new-data-at-wco-iof-esceo-2019-revealing-osteoporosis-treatment-gap-in-europe/
Accessed 20 November 2019.
15 Keshishian A, Boytsox N, Burge R, et al. Examining the effect of medication adherence on risk of subsequent fracture among women with a fragility fracture in the US Medicare Population. J Manag Care Spec Pharm 2017;23(11):1178-1190. doi: org10.18553jmcp.2017.17054.
16 Fatoye F, Smith P, Gebrye T, et al. Real-world persistence and adherence with oral bisphosphonates for osteoporosis: a systematic review. BMJ Open 2019;9:e027049. doi: 10.1136/bmjopen-2018-027049.
17 NOF 2019. National Osteoporosis Foundation. Medicare costs of osteoporotic fractures. https://www.bonehealthpolicyinstitute.org/full-milliman-report Accessed 24 November 2019.
18 Veradigm 2019. Evalytica. https://veradigm.com/evalytica-real-world-evidence-analytics/
Accessed 15 December 2019.
19 Oguntuga A, Wilk A, Tyagi G, et al. Extraction of DEXA lab values from SOAP notes using the MITRE identification scrubber toolkit (MIST) to perform named entity recognition. AMIA 2018 annual symposium. https://www.amia.org/amia2018/posters
Accessed 15 December 2019.
20 NOF 2019. National Osteoporosis Foundation. Health Professionals Toolkit. https://static1.squarespace.com/static/5d7aabc5368b54332c55df72/t/5dd2e2a92e1e1821e328308e/1574101724294/HCP+Toolkit-with+graphics.pdf Accessed 1 December 2019.
21 Chen J, Druhl E, Polepalli B, et al. A natural language processing system that links medical terms in electronic health record notes to lay definitions: system development using physician reviews. JMIR 2018;20(1):e26. doi: 10.2196/jmir.8669.
22 HealthPayerIntelligence. Using technology to close care gaps, improve care quality and cost. https://healthpayerintelligence.com/news/using-technology-to-close-care-gaps-improve-care-quality-and-cost
Accessed 15 December 2019.
23 Amgen 2019. Four Ways Amgen Is Applying AI to Combat Serious Illness. https://www.wired.com/wiredinsider/2019/06/four-ways-amgen-applying-ai-combat-serious-illness/
Accessed 15 November 2019.
24 Amgen 2019. Artificial Intelligence: A Developing Capability at Amgen.
https://www.amgen.com/media/featured-news/2018/05/artificial-intelligence—a-developing-story-at-amgen/
Accessed 15 December 2019.